| |
 |
 |
| |
| Home | Project | People | Research | Education | Archive | |
 |
Pharmacogenomics |
[English | Japanese ] |
 |
 |
  |
 |
Ken-ichi Inui
Professor,
Department of Pharmacy,
Kyoto University Hospital |
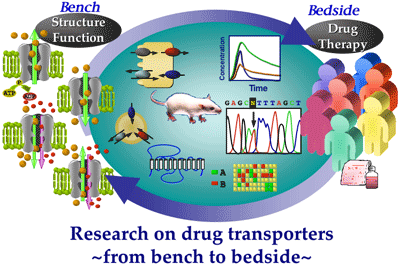
Laboratory and Clinical Studies of the Pharmacokinetics, Effectiveness and Toxicity of Drugs
We have systematically developed pharmacokinetic research from drug transport analyses based on the molecular levels to their clinical applications, i.e., functional and molecular characterization of drug transporters expressed in the intestine, kidney and liver, and the establishment of the personalized medicine based on the molecular information of drug transporters.
Tacrolimus has been widely used in many types of allograft recipients, but this agent features a wide intra- and interindividual pharmacokinetic variability, especially of bioavailability after oral administration. To establish the personalized immunosuppressant therapy, the relationship between the tacrolimus concentration/dose (C/D) ratio and the intestinal mRNA expression level of MDR1 or CYP3A4 in the recipients of living-donor liver transplantation (LDLT) were investigated. As a result, we have demonstrated that the intra- and interindividual variability of tacrolimus pharmacokinetics in allograft recipients is related to the intestinal expression level of MDR1, but not that of CYP3A4. This information is utilized for the determination of the initial dosage regimen of tacrolimus for LDLT recipients. For many drugs excreted into the urine, dosage regimens are usually adjusted according to creatinine clearance. However, creatinine clearance reflects only the glomerular filtration rate of normal kidney, and dosage schedule based on the creatinine clearance was reported to be inadequate for many drugs. To overcome this clinical problem, we have pursued the novel biomarker for renal drug excretion, and found that mRNA expression levels of organic anion transporter 3 (OAT3/SLC22A8) were well correlated with renal secretion of an anionic cephalosporin antibiotic cefazolin. Thus, the expression levels of renal drug transporters are suggested to be a useful marker to evaluate the renal function.
We have performed following research projects:
1) Functional and molecular characterization of drug transporters and its clinical application,
2) Pathophysiological roles of drug transporters,
3) Mechanisms of drug interaction,
4) Pharmacogenomics and personalized medicine.
 |
Top
| 2007 | 2006 | 2005 | |
|
|

